The FDA has requested an extension of “up to three months” in order to complete the review of the NovaDel’s Zolpimist Oral Spray for the short-term treatment of insomnia. The FDA will reply on or before December 19, 2008.
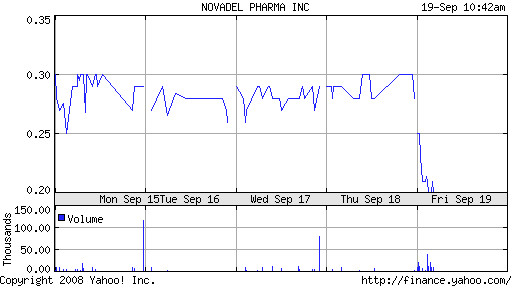
NovaDel shares, which opened at $.28, traded as low as $.20 today.
Following NovaDel’s regulatory strategy, the company submitted the NDA using the 505(b) (2) path. This allows a NovaDel to rely upon the FDA’s findings of safety and efficacy for a previously approved drug, shortening the development time.
NovaDel’s application is based on data from two randomized, open-label, dose-ranging studies comparing ZolpiMist with Ambien tablets in young and elderly healthy volunteers. Both studies compared the pharmacokinetics and safety of comparable doses of zolpidem administered as an oral spray versus tablets. The speed of drug absorption and level of sedation were also assessed. The results demonstrated bioequivalence between ZolpiMist and Ambien.
To avoid a repeat of the ill-fated Hana Biosciences’ partnership, NovaDel also included data from process validation and registration stability batches produced at the intended commercial manufacturing facility.
The U.S. patent for zolpidem was held by the French pharmaceutical giant Sanofi-Aventis until April 2007 when the FDA approved 13 generic versions of zolpidem tartrate. Zolpimist is a pending trademark of NovaDel.
NovaDel’s Spray formulations contain the drug in solution, ready to be absorbed through the oral mucosa directly into systemic circulation. When oral spray is utilized the drug can potentially reach effective blood levels in as little as 10-15 minutes. In bypassing the GI tract and liver, the same therapeutic benefits can be achieved more quickly. The spray formulation should provide faster relief of sleeplessness; it may also be possible to achieve the same effect with a lower dose and improve the side effect profile.